One of the first questions people ask is about the science behind how carbon from ashes becomes a diamond. They wonder, how much carbon is in the human body after cremation? Is there really enough left in the ashes to create a diamond? The answer is a resounding yes. As carbon-based lifeforms, 18.5% of our body is made of this essential element. It absolutely survives the cremation process, and there’s more than enough to begin the remarkable carbon to diamond journey.
While some of that carbon is transformed into carbon dioxide during cremation, 1-4% still exists in the ashes and fragments of bones that remain after the process, yielding an average of 2.5 to 8.5 mg of carbon. Since we only need 12 mg of carbon to grow a diamond, there is more than enough to facilitate the growth process of multiple diamonds with only a half cup of ashes.
In this article, we’re going to take a closer look at the science behind extracting carbon from cremated remains and transforming it into lab-grown diamonds in order to provide clarity regarding the process and confidence to those who want to memorialize their loved ones through this journey.
So, How Much Carbon Is Actually in Your Body?
There are a total of 26 unique elements found in the human body, 11 of which are found in quantities above 0.01%. Of those 11 primary elements, there are four key substances that make up 96.2% of the average person, including:
· Oxygen (O): 65%
· Carbon (C): 18.5%
· Hydrogen (H): 9.5%
· Nitrogen (N): 3.2%
Carbon is a fundamental building block of life, plays a major part in the formation of DNA, and can be found throughout the body in various bonded compounds that form our bones, muscle tissue, major organs, skin, and hair.
During the cremation process, a body is exposed to temperatures ranging from 1,400°F to 1,800°F, transforming much of the solid matter to gasses through combustion. One of these gasses is carbon dioxide, which takes with it a large percentage of the body’s carbon. However, after cremation, bone fragments and other base elements remain, which are then reduced to the powdered ashes that are returned to the family.
Bones consist mainly of water, carbon-based tissues in the form of protein collagen, and other minerals. Cremation burns off the water and the majority of the living tissue, but the remaining minerals, which include a type of calcium phosphate known as hydroxyapatite, contain carbonates—a source of carbon. So, while the amount of remaining Carbon is greatly reduced from 18.5% to roughly 1-4%, there is still a significant amount present in the material left behind after cremation.
With this material, scientists have been able to use carbon-dating on cremated remains since 2001, and the existence of residual carbonates in cremated remains was later validated by a study conducted in 2009 by the Department of Biology and Department of Anthropology at Ludwig-Maximilian-University Munich as well as other researchers from accredited schools of anthropology and forensics such as University of Cambridge in 2016.
In the 2009 study, researchers collected cremated remains for analysis and cremated the tibia bone of a cow at temperatures ranging from 212°F to 1,832°F to compare the reduction of the remaining carbonates as the temperature was increased. Each piece of bone used in the study was held at the control temperature for one and a half hours and then tested to determine how its elemental composition was affected by the increasing cremation temperature.
Researchers found that there were residual carbonates remaining in the test samples and cremated remains all the way up to the maximum temperature above 1,832°F, confirming that some amount of carbon would survive even the highest cremation temperatures.
“There are residual Carbonates all the way up to the maximum cremation legal temperatures above 1,800°F.” - Forensic Science International
The Role of Water
It’s incredible to think that our bodies are mostly water. According to the Mayo Clinic, water makes up 50% to 70% of our total body weight and is essential for every single cell, tissue, and organ. This high water content is a key reason why the cremation process results in a much smaller volume of remains. During cremation, this water is evaporated, leaving behind the core mineral and elemental components of the body. Understanding this helps clarify how the physical form changes, while the fundamental elements, like carbon, remain concentrated in the ashes that are returned to the family.
The Six Main Building Blocks
When you look past the water, our bodies are composed of a few key elements that form the very foundation of our existence. The National Institute of General Medical Sciences notes that about 96% of our bodies are made of just four main elements: oxygen (65%), carbon (18.5%), hydrogen (9.5%), and nitrogen (3.3%). The other two primary elements are calcium and phosphorus. Carbon’s significant presence, at 18.5%, is what makes us carbon-based lifeforms. It's this specific element that we are able to isolate and use to create a beautiful and lasting memorial. This core component of life provides the essential material needed for the entire diamond-growing journey.
Essential Secondary and Trace Elements
While the main elements get most of the attention, the remaining 4% of our bodies are just as important. This portion is made up of secondary and trace elements, many of which are metals that play crucial roles in our biological processes. These include elements like potassium, sulfur, sodium, chlorine, and magnesium. The unique combination of these trace elements is part of what makes every person’s chemical signature distinct. During the carbon purification process, many of these other elements are removed, but their unique presence in the initial ashes contributes to the one-of-a-kind nature of your loved one’s final memorial diamond.
Other Interesting Facts About Carbon
Carbon is truly the stuff of life. It’s not just a number on the periodic table; it’s a fundamental building block that forms our DNA and is found in the bonded compounds that create our bones, muscles, organs, skin, and hair. Because carbon is so integral to our hair, it’s also possible to create a diamond from hair samples, offering a way to celebrate a living person or to memorialize a loved one if ashes are not available. This versatility highlights just how central carbon is to our identity. It’s the element that connects every part of us, and it’s the very same element that can be transformed into a timeless symbol of remembrance.
The Function of Key Elements in the Body
While carbon gets a lot of the spotlight for being a fundamental building block of life, it’s just one member of a whole team of elements working together. Think of your loved one’s body as a complex and beautiful symphony, where each element played a crucial part in the music of their life. These chemical elements are the basic building blocks for everything from DNA to organs. Understanding the roles of these other key players gives us a deeper appreciation for the intricate science that made your loved one so uniquely them.
Calcium and Phosphorus: The Structural Duo
Calcium and phosphorus are the primary architects of the human body, forming the strong framework of our bones and teeth. Bones are more than just a scaffold; they are a living tissue composed of protein collagen and minerals, primarily a type of calcium phosphate. It’s within this mineral structure that carbonates—a key source of carbon—are stored. This is why even after cremation, enough essential carbon remains within the bone fragments to create a memorial diamond. These two elements provided the physical strength and structure that carried your loved one through every step of their journey.
Potassium and Sodium: The Nerve Communicators
If calcium and phosphorus are the framework, potassium and sodium are the body’s intricate communication network. These elements work together to create the electrical impulses that allow nerves to fire and muscles to contract. They powered every thought, every heartbeat, and every message sent from the brain to the rest of the body. This delicate balance of potassium and sodium was essential for the electrical energy that animated your loved one, enabling them to experience the world, share a laugh, or give a warm hug. They were the spark behind the connection you shared.
Sulfur and Magnesium: The Protein and Enzyme Helpers
Sulfur and magnesium are the unsung heroes, working diligently behind the scenes. Sulfur is a key component of essential amino acids, which are the building blocks of proteins that form everything from hair and skin to muscle tissue. Magnesium, on the other hand, is a helper for hundreds of enzymes, acting as a catalyst for countless biochemical reactions that sustain life. Together, they ensured all the microscopic machinery in your loved one’s body ran smoothly, contributing to their health and vitality in ways both seen and unseen.
Iron: The Oxygen Transporter
While it makes up only a tiny fraction of the body’s mass, iron has one of the most critical jobs: transporting oxygen. Primarily found in the hemoglobin of red blood cells, iron is what captures oxygen in the lungs and delivers it to every single cell. This process is what fueled your loved one’s energy and sustained their life force. Although about 96% of our bodies are made of oxygen, carbon, hydrogen, and nitrogen, it’s this small but mighty amount of iron that ensures the life-giving oxygen reaches its destination, coloring the blood and, in a way, coloring our lives.
Are There Harmful Elements in the Body?
It’s a valid question to wonder if other, less desirable elements are present in the body. Over a lifetime, it’s natural to accumulate trace amounts of harmful elements like lead, mercury, or cadmium from our environment. While these can have toxic effects on health, their presence in trace amounts is a normal part of the elemental makeup of any modern person. When considering a memorial diamond, it’s important to know how these elements are handled. The beauty of the diamond creation process is that it’s fundamentally a process of purification.
Here at Eterneva, our first step is to isolate and purify the carbon from all the other elements present in the ashes. Our highly scientific method is designed to extract only the essential carbon, leaving behind any impurities, including heavy metals and other trace elements. This ensures that the foundation of your loved one’s diamond is nothing but their pure, unique carbon signature. Our commitment to transparency and scientific integrity means that the final gem is a brilliant and uncontaminated celebration of the life it represents, capturing their pure essence forever.
Is There Enough Carbon Left in Ashes for a Diamond?
Carbon testing is a standard practice used for many different purposes in a variety of industries. The most commonly known type of carbon testing is known as carbon dating, used by scientists to determine the age of organic matter through the relative proportions of carbon isotopes contained within it.
Another kind of carbon testing, known as total carbon analysis (TC), is used in common industries like pharmaceuticals, microelectronics, oil and gas, and forensics. Total carbon analysis utilizes scientific methods to test specifically for both organic and inorganic carbon
A sample is placed in an oxygen-rich controlled environment and combusted (burned) with a quartz heating element, allowing testing to proceed without adding any extra fuels or additives that could contaminate the testing sample. After combustion in the controlled chamber, carbon combines with oxygen to create carbon dioxide, which is then analyzed with an infrared detector to determine the quantity of Carbon in the sample.
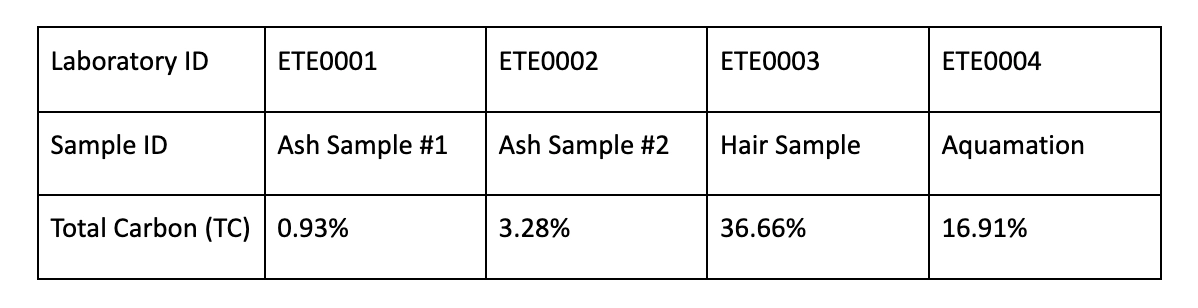
Eterneva used this same TC technique on samples of cremated ashes, hair, and even aquamation (eco-friendly cremation method) ashes to determine the range of carbon percentage we can expect from each. All testing was performed on controlled samples at independent environmental laboratories. These were the results:
Once the carbon levels of the ashes have been determined and are sufficient, the scientists at Eterneva can begin the process of transforming the carbon into a diamond.
How Much Carbon is Needed for a Memorial Diamond?
12 mg of carbon (approximately 0.06 carats) is needed to make a memorial diamond. To ensure you'll have enough carbon from cremated ashes, you would need half a cup of ashes.
Read our ashes to diamonds guide on the number of memorial diamonds you can create to learn more.
The First Step: Purifying Carbon from Ashes
While carbon exists in the molecular form of a carbonate compound, carbonates must first be decomposed using a high heat reduction process to purify the cremated remains into a form of carbon graphite that can be used to grow a diamond. This is achieved in a two-part process set in an extremely high-temperature, low-oxygen controlled environment.

This complex method requires highly specialized equipment and high-level expertise but is capable of reducing organic and inorganic materials containing carbonates and free-form carbon to crystalline graphite powder. First, gasses are used to separate impurities in the carbonates, then the remaining graphite is heated to more than 4892 °C, causing more impurities with low boiling points to vaporize and are removed. This process results in graphite (pure carbon) that is more than 99.995% pure and ready to use.
How That Carbon Becomes a Diamond
The use of High Pressure High Temperature (HPHT) technology to grow diamonds in a lab originated in the 1950s and was pioneered in the United States by General Electric. While the technology’s origins were centered on industrial applications, the capability to grow jewelry-grade diamonds has advanced greatly in recent years. Since the early 1990s, lab-grown diamonds have been produced with increasing size and quality to the point where they are no longer graded any differently than natural diamonds, and provide a comparable alternative to their mined counterparts.
In general, HPHT growth begins with recreating the temperatures, pressures, and conditions similar to those that occur in the mantle of the earth during natural diamond growth. Diamonds typically form at pressures of 5.5–8.0 GPa (roughly the weight of a commercial jet plane balanced on a fingertip!) and temperatures of 2,500-3200°F, deep within the earth.
To mimic this, HPHT diamond growth involves placing the carbon graphite powder into a growth cell and exposing it to similarly immense pressures and temperatures. The HPHT growth cell is made up of a metal alloy, which helps to facilitate diamond growth at a lower temperature than is required in nature. It also includes a small diamond seed the size of a grain of sand that functions as the formation site where the carbon can attach and begin its growth into a full diamond.

A memorial diamond is a mix of personal carbon extracted from ashes and generic carbon that crystalizes around a tiny diamond seed to form a beautiful diamond. Both the personal carbon derived from a person’s cremated ashes and the generic carbon undergo the same HPHT process described above.
Due to the existence of minuscule impurities that tend to exist in carbon from cremated ashes, even after purification, which can affect the clarity and growth of the diamond, the personal carbon is blended with additional generic carbon. Personal carbon makes up 10-15% of the carbon used to create the diamond, But by introducing additional generic carbon, the HPHT technology is able to grow a diamond of exquisite quality, brilliance, and depth, while still encompassing the remarkable and unique life of the loved one encompassed by it.
At Eterneva, we recognize carbon for the incredible material it is—one of the building blocks of Earth and a fundamental substance in our universe. It is the chemical backbone of organic life on Earth, a cosmically connective element that can be found in stars, planets, the ocean, and our atmosphere. It is in our loved ones and in ourselves, and it carries on after we have passed.
To use it to create a beautiful memorial diamond can be a transformative and powerful process, reminding us of our connections, our memories, and the cosmic cycle of our lives.
Frequently Asked Questions
How can you be sure there's enough carbon left in ashes after cremation? It’s a great question, and the science is fascinating. Our bodies are about 18.5% carbon, and while the high heat of cremation transforms a lot of it, a significant amount remains locked within the bone fragments as carbonates. Scientific studies have confirmed this, showing that even at the highest cremation temperatures, there is more than enough of your loved one's unique carbon left to create a beautiful diamond.
Is the final diamond made 100% from my loved one's carbon? The diamond is a blend of your loved one's personal carbon and additional generic carbon. We do this to ensure the final gem grows with the highest quality, clarity, and brilliance possible. Your loved one's carbon is the essential foundation, making up about 10-15% of the total, which ensures their unique essence is at the very heart of the diamond that is grown just for them.
What happens to all the other elements in the ashes during this process? The very first step in our journey is purification. We use a highly scientific process to isolate only the carbon from the ashes. This means all the other elements, like calcium, phosphorus, and any trace metals, are carefully removed. This step ensures that the foundation of your diamond is nothing but pure carbon, resulting in a brilliant and flawless gem that honors your loved one.
Can I use hair instead of ashes to create a diamond? Yes, you absolutely can. Just like ashes, hair is very rich in carbon, which is the fundamental element needed to grow a diamond. This makes it a wonderful option for creating a living tribute to celebrate someone special, or as an alternative if you don't have access to ashes. The process is very similar and results in an equally meaningful and beautiful diamond.
How much ash is actually needed to start the process? We only need about a half cup of ashes to begin. It might not seem like a lot, but that small amount contains more than enough of the essential carbon required for the entire diamond-growing journey. This allows you to keep the majority of your loved one's remains for other memorials if you wish.
Key Takeaways
- Your Loved One’s Carbon Endures: Our bodies are rich in carbon, and science confirms that more than enough of this essential element remains in ashes after cremation to create a beautiful and personal memorial diamond.
- Purity Is the First Step: The journey from ashes to diamond begins with a meticulous scientific process that isolates your loved one's unique carbon, removing all other elements to create a pure foundation for the gem.
- Technology Recreates Nature's Process: Using advanced High Pressure High Temperature (HPHT) methods, the purified carbon is placed in an environment that mimics the earth's natural forces, transforming it into a brilliant, genuine diamond.













